Absorption and light intensity equation4/23/2024  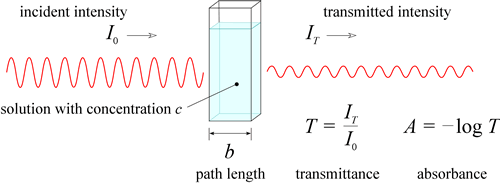
It is also important to note the strength of absorbance at these wavelengths.īy looking at these peaks, you can establish the molar attenuation coefficient, ε. Identifying these wavelengths can can help you determine certain molecular properties of your sample. When you measure the absorbance spectrum of a solution, it is important to at which wavelengths maximum absorbance occurs. This absorbance, A, can be defined in a number of different ways for different samples. Defining AbsorbanceĪbsorbance is a relative measurement so is therefore unitless. You can calculate absorbance by the comparing the reference light spectrum, I 0, tothe detected light that has passed through the sample, I, through this equation. Light is transmitted through the sample and recorded by the spectrometerĪs light is propagated through a sample and absorbed, its intensity will fall exponentially with distance. Absorbance values are closely related to transmission measurements, as in both cases you actually measure the transmitted light. To measure absorbance requires a broadband light source and a spectrometer with appropriate spectral range. For example, the types of electronic transitions that are allowed will often vary with different material properties. Measuring the wavelengths at which absorbance occurs will therefore tell you a lot about the electronic properties of a material, molecule or thin film. When multiple atoms come together to form molecules, energy levels become more complex. The wavelengths of the absorbed light correspond to energy through the equation. However, if the photon is less than this energy, it will not be absorbed. 
If the photon is E ph > ΔE, an electron will absorb the photon, and it can be excited from the HOMO to the LUMO. Photon absorbance in a molecule with energy gap, ΔE. If the photon energy is high enough, it can be absorbed by an electron in the HOMO and will be excited into a higher energy state. The wavelength of light that can be absorbed by a material is dependent on these different energy levels and the electronic distribution within this material.Ībsorption occurs when the the energy of the incoming photon is equal or larger than energy difference (ΔE) between the highest occupied molecular orbit (HOMO) and the lowest unoccupied molecular orbit (LUMO). Depending on the electronic structure of an atom, these orbitals (or energy levels) can be filled, partially filled, or empty. Within an atom, electrons can exist in regions around the nucleus known as orbitals. This can be useful when discussing different properties of a solid (conductors, semiconductors, or metals). See also: Absorption spectrometry: using measurements at different light frequencies.Band theory describes how electrons are organised within a solid. The quantity is called the coefficient of absorption of the -th gas, and can be measured in the laboratory. The coefficients, correspond to the log-ratio of light intensities when (the -th vector of the standard basis, which correspond to the -th pure gas). The log-ratio of intensities is thus of the form for some vector, where is the vector of concentrations. If the container has a mixture of ‘‘pure’’ gases in it, the law postulates that the logarithm of the ratio of the light intensities is a linear function of the concentrations of each gas in the mix. In other words,, where the constant depends on the light frequency and on the gas. The Beer-Lambert law postulates that the log-ratio is linear in the concentration. Depending on the absorption properties, as well as the concentration, of the added gas, the light will be more or less absorbed with respect to the reference situation. 
Then, we add some other gas to the container, repeat the experiment, and measure the light intensity again (say, ). We measure the light intensity (say, ) at the other side of the container. First we do a control experiment, where we illuminate from one side a container containing some reference gas with light at a certain frequency. The principle of an absorbtion spectrometer, illustrated on the left, is as follows. This is the basis of absorption spectrometry, which allows to measure the concentration of different gases in a chamber. The Beer-Lambert law in optics is an empirical relationship that relates the absorption of light by a material, to the properties of the material through which the light is travelling. Beer-Lambert Law in Absorption Spectrometry 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
