The halogens are a series of non-metal elements from group 17 of the periodic table (formerly VII). They share similar chemical properties.ĮlectronegativityThe tendency of an atom to attract electrons to itself. HalogensGroup 17 (or VII) in the periodic table consisting of fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). Halogens can be harmful or lethal to biological organisms in sufficient quantities. Therefore, they are highly reactive and can gain an electron through reaction with other elements. Therefore, the physical state of the elements down the group changes from gaseous fluorine to solid iodine.ĭue to their high effective nuclear charge, halogens are highly electronegative. As a diatomic molecule, fluorine has the weakest bond due to repulsion between electrons of the small atoms.ĭue to increased strength of Van der Waals forces down the group, the boiling points of halogens increase. Halogens are nonmetals in group 17 (or VII) of the periodic table. Iodine and bromine are added to halogen lamps to Most nonmetals has led to a wide variety of uses for these elements.Ĭhlorine is used as a bleach and a disinfectant. The ability of halogens to form chemical compounds with all metals and Number of –1, whereas the other halogens can exhibit a range of Halogens are so reactive that all the elements except helium and neon haveīeen found to react with at least one of the halogens. Fluorine has the highest electronegativity of all the elements. Remove an electron from an atom of another element. These ions have a closed outer-shell configuration.Įlectronegativity is a measure of the ability of an atom of one element to AnĪdditional electron can be added to halogen atoms to form singly charged The neutral atoms of the halogens possess seven outer electrons. Halogens have distinctive, unpleasant odors, will burn exposed flesh, and The halogens are poor thermal and electricalĬonductors in all phases, and as solids they are brittle and crumbly. Solid iodine appears as shiny, dark gray crystals, and the Liquid bromine and its vapors areīrownish red. 
Fluorine gas is pale yellow,Īnd chlorine gas is a yellowish green. All of the elements are colored, with the colorīecoming more intense moving down the group. Physical states: Fluorine and chlorine are gases, bromine is a liquid, and Near room temperature, the halogens span all of the They have relatively low melting and boiling points that increase steadilyĭown the group. Halogens display physical and chemical properties typical of nonmetals. Salts (halides) as sodium chloride (NaCl) and potassium iodide (KI). The salinity of the oceans on Earth is due in large part to such halogen The family name means "salt-forming," from the Greek for High reactivity, the halogens are never found in nature in native form. Elemental halogens are diatomic molecules. The halogens make up Group VIIA of the Periodic Table of theĮlements. (atomic symbol F), chlorine (Cl), bromine (Br), iodine (I), and astatine Chlorine is less reactive and much more manageable, and added to water in small quantities to kill microorganisms to make it safe to drink.The halogens are the family of chemical elements that includes fluorine Fluorine is so eager to react with anything that it is almost never found as a pure element and it is so dangerous to work with that scientists avoid handling it in reactivity experiments. This means that fluorine, at the top of the group, is the most reactive. Unlike the group 1 metals, reactivity decreases as you go down the halogens. Iodine is a dark grey solid which gives off a purple vapour when heated 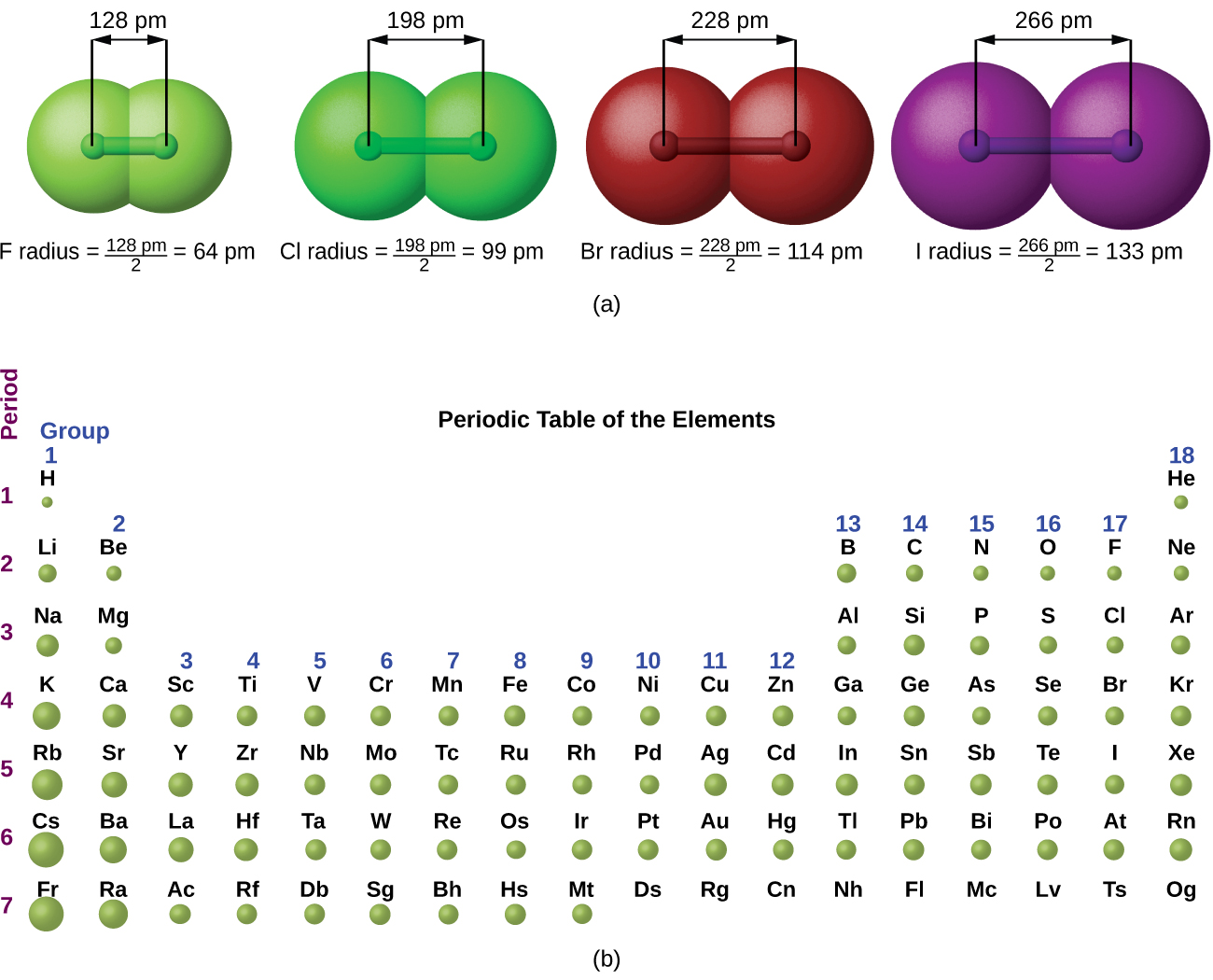
Each halogen has the following characteristics at room temperature: The more electrons an element has, the more intermolecular forces (London dispersion forces) it can form. Boiling point increases as you go down the group because the mass of each element increases and they have more electrons around their nuclei. As you go down the halogens, from fluorine to astatine, the elements become darker in colour and have a higher boiling point. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
